Localised prostate cancer is cancer that has not spread outside the prostate gland, often referred to as organ-confined (stage T1/T2). It is often slow-growing, has a high likelihood of recovery, and may not require immediate treatment, potentially allowing for active surveillance, though it can also be treated with surgery or radiation.
Treatment options for localised prostate cancer include active surveillance, surgery or radiation therapy.
Active Surveillance is a reasonable option for low risk localised prostate cancers.
Localised Prostate Cancer
Staging of prostate cancer uses the TNM staging to determine localisation whether organ confined (localised disease) or spread outside the prostatic capsule (locally advanced disease) or distance spread (advanced disease or metastatic disease).
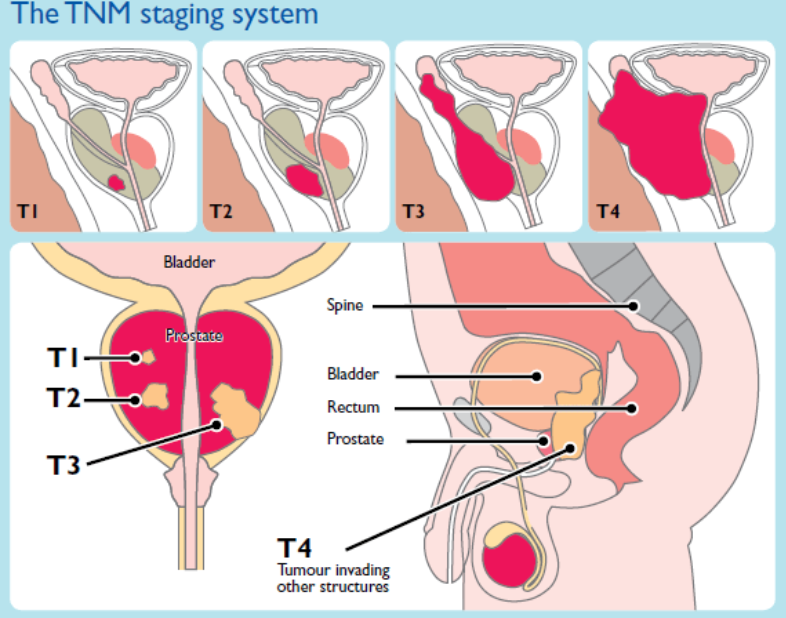
TNM Staging Details for Prostate Cancer
T – Primary Tumor (Clinical staging via MRI, DRE, and biopsy):
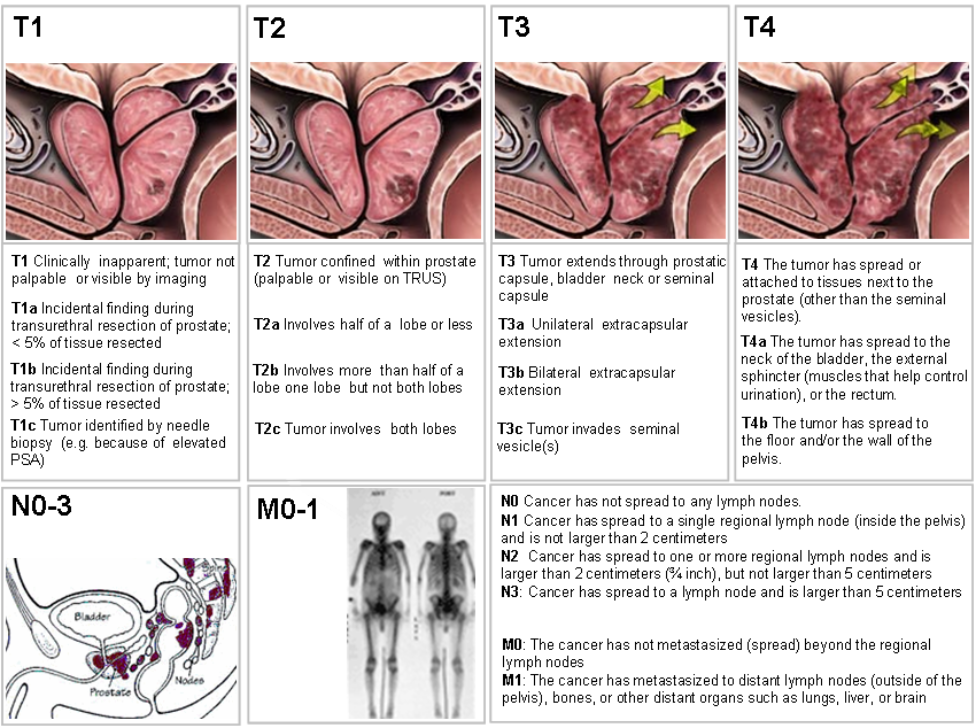
T1: Clinically inapparent tumor, not palpable, typically found in a biopsy.
T1a: Found in 5% or less of resected tissue.
T1b: Found in more than 5% of resected tissue.
T1c: Found by needle biopsy but not palpable.
T2: Tumor is palpable and confined within the prostate.
T2a: Involves one-half of one side (lobe) or less.
T2b: Involves more than one-half of one side, but not both.
T2c: Involves both sides.
T3: Cancer has spread through the capsule (T3a) or into the seminal vesicles (T3b).
T4: Tumor has invaded adjacent structures other than the seminal vesicles, such as the bladder, rectum, or pelvic wall.
N – Regional Lymph Nodes:
N0: No spread to regional lymph nodes.
N1: Cancer has spread to regional lymph nodes (within the pelvis).
M – Distant Metastasis:
M0: No distant metastasis.
M1: Distant metastasis is present.
M1a: Non-regional lymph nodes.
M1b: Bone metastasis.
M1c: Other sites (e.g., liver, brain, lungs)
Localised prostate cancer is said to occur when the cancer is contained entirely within the prostate gland, having not spread to the capsule, nearby tissues, or distant parts of the body. This corresponds to clinical T1 or T2 stages, meaning the cancer has not spread beyond the prostatic capsule.
Localised prostate cancer is usually asymptomatic (without any symptoms) and at times present with early urinary symptoms. Localised prostate cancer is detected through staging of the disease via imaging after histopathological confirmation of prostate cancer. Treatment options include active surveillance, surgery, or radiation, with high survival rates.
Active surveillance is a management strategy for low-risk, early-stage prostate cancer where the disease is closely monitored with regular tests instead of being treated immediately with surgery or radiation. The primary goal is to delay or avoid treatment-related side effects while ensuring that any cancer growth is caught early enough for curative treatment.
Risk Stratification Of Prostate Cancer And What Is Low Risk Prostate Cancer
Risk stratification of prostate cancer is the process of categorising patients into groups based on the clinical “aggressiveness” of their disease. This helps decide whether a patient needs immediate treatment (like surgery or radiation) or can be safely monitored through active surveillance.
Risk stratification is critical to determining the aggressiveness of prostate cancer and consequently the treatment plan. It is based on three main factors: PSA level, Gleason score, and clinical stage (T stage).
Core Factors Used in Stratification
Most traditional models use three primary pieces of information to determine a patient’s risk group:
- PSA Level: The amount of prostate-specific antigen in the blood. Higher levels (especially >20 ng/mL) indicate higher risk.
- Gleason Score / Grade Group: A pathological assessment of how abnormal the cancer cells look under a microscope. Grade Group 1 is the least aggressive, while Grade Group 5 is the most.
- Clinical Stage (TNM): Based on a physical digital rectal exam (DRE) or imaging to see if the tumour is contained within the prostate or has spread.
- Percentage of Positive Cores: Often defined by low-volume disease (e.g., fewer than three biopsy cores are positive) or high volume disease.
Standard Risk Groups
While different organisations (like the NCCN or EAU) have slightly different criteria, patients are generally grouped as follows:
- Low Risk: PSA <10 ng/mL, Grade Group 1 (Gleason 6 = 3+3), and stage T1-T2a. These cancers are unlikely to spread quickly.
- Intermediate Risk: PSA 10–20 ng/mL or Grade Group 2-3 (Gleason 7 = 3+4, 4+3). This is often split into favorable (Gleason 3+4) and unfavorable subgroups (Gleason 4+3) to refine treatment choices.
- High Risk: PSA >20 ng/mL, Grade Group 4-5 (Gleason 8–10), or evidence the cancer has grown outside the prostate (Stage T3). These require more intensive, often multimodal treatment.
Modern Enhancements
To improve accuracy beyond the basics, currently in 2026 clinicians increasingly use:
- Multiparametric MRI (mpMRI): Provides detailed anatomical data to identify suspicious lesions and accurately stage the disease.
- Genomic Testing: Tests like Oncotype DX or Prolaris analyze the genetic “signature” of the tumour to predict its biological behaviour.
- Cambridge Prognostic Groups (CPG): A newer 5-tier system increasingly used (especially in the UK by NICE) that better distinguishes between different levels of intermediate and high risk
Low-risk prostate cancer
Low-risk prostate cancer implies the cancer is unlikely to spread quickly. To be considered low-risk, the cancer must meet all of the following criteria:
- Grade Group 1: The Gleason score is 6 (3+3).
- PSA Level: Less than 10 ng/mL.
- Clinical Stage T1–T2a: The tumor is not palpable (T1) or can be felt but is confined to less than half of one side (lobe) of the prostate.
- Percentage of Positive Cores: Often defined by low-volume disease (e.g., fewer than three biopsy cores are positive).
“Very low-risk” prostate cancer is a subset of low risk prostate cancer defined by smaller tumor volume and lower PSA density (PSA density lower than 0.15 ng/mL/mL).
Active Surveillance
Prostate cancer is the poster-child example of a scrutiny-dependent cancer – meaning the more one screens, the more number of cancers will be detected.
The reason active surveillance is an option for low risk early stage prostate cancer is because most patients with prostate cancers are slow-growing (indolent) and never cause symptoms or death, leading to the saying that most men die WITH prostate cancer rather than OF it.
The “Whitmore Principle,” famously phrased by Willet Whitmore states: “For a patient with prostate cancer, if treatment for cure is necessary, is it possible? If possible, is it necessary?” Fortunately, most men die with prostate cancer, not of it.
The specific characteristics of prostate cancers, unlike other cancers, are:
1. Prostate cancer is a scrutiny-dependent cancer. The more one screens and diagnoses, the more cases will be detected. But this does not mean that early detection will improve cancer-specific mortality.
2. Prostate cancer has a long mean sojourn time, meaning it often exists in a slow, asymptomatic pre-clinical phase for many years (often a decade or more) before it’s detectable by symptoms or routine screening, allowing for early detection but also raising concerns about over-diagnosis and overtreatment of slow-growing cancers that might never harm a man, even if found early.
Low-risk, localised prostate cancer is typically very slow-growing, often taking 10 to 15 years or longer to spread or cause significant health issues. For many, it may never progress to a fatal stage, with a 10-year survival rate of approximately 99%. It often poses a lower risk of mortality than other health conditions in elderly patients.
Nearly 100% of men with localised, low-risk prostate cancer survive 5 years or more after diagnosis and hence do not require active treatment.
Because of its slow progression, Active Surveillance is often recommended for low risk early stage localised prostate cancer, involving monitoring rather than immediate surgery, radiation therapy or any other modalities of intervention.
While uncommon for localised prostate cancers to spread rapidly, if the cancer is actually more aggressive (e.g., Gleason 7-10), it can spread to bones or other organs in a few years. Hence Gleason 7-10 prostate cancers are not consider low risk cancers
Risk Factors for Faster Progression: A rapidly rising PSA level particularly if the doubling time is less than 12 months, or a high initial Gleason score (aggressive grade of the cancer) indicates a higher likelihood of faster progression. Hence these categories are not considered low risk, and are not subjected to the option of Active Surveillance.
Active Surveillance is typically recommended for men with small tumors that are confined to the prostate and are expected to grow very slowly. Ideal candidates generally have:
- Gleason Score: 6 or lower (sometimes a “favorable” 7 = 3+4).
- PSA Level: Usually 10 ng/mL or lower.
- Stage: Localized (T1 or T2 stage).
- Symptoms: No significant symptoms or signs of the cancer spreading.
What Active Surveillance Involves
A typical surveillance schedule includes a series of regular tests at predefined periods of time:
- PSA Blood Tests: Done every 3 to 6 months to check for rising levels.
- Digital Rectal Exams (DRE): Performed once or twice a year to feel for changes in the prostate’s surface.
- Imaging (MRI): Often used every 1 to 3 years to track the size and location of the tumor.
- Repeat Biopsies: Usually done every 1 to 5 years to re-evaluate the cancer’s grade and aggressiveness.
Active Surveillance Versus Watchful Waiting
While both involve observing or watching the cancer, they are distinct approaches:
Active Surveillance is aimed at curing the cancer later with an active intervention with surgery or radiation therapy if it subsequently progresses while on active surveillance. This involves intensive, regular testing (including serial PSA tests, MRI, prostate biopsies).
Watchful Waiting is generally for men with a shorter life expectancy or other serious health issues. It involves fewer tests and focuses on managing symptoms rather than curing the disease.
During active surveillance, if test results show significant changes – such as a rapidly rising PSA or a higher Gleason score on a repeat biopsy, the urologist will typically recommend moving to active treatment options like surgery or radiation therapy. But this has to be critically evaluated based on the predicted residual life expectancy of a patient under surveillance. If there appears to be a rising PSA when a patient has already reached let’s say 80 years of age, and he is already reaching his last days of life expectancy anyway, he would be switched to watchful waiting rather than an active intervention for prostate cancer.
Verdict in 2026
As of early 2026, the clinical verdict on Active Surveillance for prostate cancer is that it remains the gold standard and preferred management strategy for low-risk and favorable intermediate-risk prostate cancer, with growing evidence supporting its long-term safety.
New data and guidelines presented in 2026 emphasize a shift toward more personalised surveillance, using advanced imaging and genomic tests to reduce over-testing while maintaining a very low risk of mortality.
Key findings from the 2026 updates include:
Long-Term Safety: Extended data from studies like the Göteborg-1 trial show that even after 25 years, prostate cancer-specific mortality for men on active surveillance remains very low.
MRI and Biomarkers as Standards: The 2026 EAU guidelines and clinical research emphasis MRI, target biopsy, and molecular markers (such as the Decipher Genomic Classifier) to refine surveillance, reducing the need for aggressive treatment in low-grade cases.
Role in Intermediate Risk: While surveillance is standard for Gleason 6 prostate cancers, it is increasingly accepted for favorable intermediate-risk patients (e.g., those with low-volume Gleason 3+4=7 disease).
“No Safe Window”: While safe, 2026 analyses stress that Active Surveillance is not “risk-free” and requires sustained monitoring to ensure patients do not miss the window of curability, with a “non-negligible” risk of disease progression, particularly over 10-15 years.
Overcoming Underutilization: Despite high efficacy, research presented at the 2026 ASCO GU symposium indicates that active surveillance is still underutilised in some demographics, particularly in younger men with very low-risk disease who often choose definitive treatment instead.
Emerging Tools: New trials are assessing the role of AI-based tools and non-invasive blood tests like the SNEP assay (Subtraction Normalised Expression of Phagocytes Blood Test) to better manage patients on active surveillance.






