External beam radiotherapy, along with radical prostatectomy (surgery), has been a mainstay treatment option for localised prostate cancer and is currently recommended by numerous guidelines for the treatment of intermediate- and high-risk localised disease.
Radiation therapy (radiotherapy) is one of the cornerstone treatment for prostate cancer, designed to cure or control the cancer by using high-energy X-rays to damage the DNA of cancer cells. Radiotherapy primarily works by using ionising radiation to induce DNA damage within cancer cells, leading to mitotic death or apoptosis, ultimately causing the tumor to shrink or cease in growth. High-energy beams (photons or protons) or internal seeds break both strands of DNA, preventing repair and triggering cancer cell death over weeks or months.
Modern radiotherapy for prostate cancer has evolved from conventional, wide-field methods to highly precise techniques that significantly reduce side effects while allowing for higher, more effective doses.
Radiotherapy for prostate cancer can either be delivered by External Beam Radiotherapy (EBRT), brachytherapy or a combination of both.
Background Knowledge In Radiation Biology
Role and significance of alpha beta ratio in prostate cancer
The use of radiotherapy in prostate cancer is heavily influenced by a biological concept known as the Alpha / Beta ratio. Understanding this ratio is essential for modern treatment because it dictates why certain scheduling methods (like 5 days of treatment vs. 8 weeks) are used.
The Alpha/Beta Ratio: Significance & Role
The alpha / beta ratio is a mathematical parameter from the Linear-Quadratic Model of cell killing that measures a tissue’s sensitivity to radiation fractionation, typically measured in Grays (Gy). It defines the dose at which the linear and quadratic components of cell killing are equal, helping determine how tissues respond to varying dose sizes. High ratios (approximately 10 Grays) indicate tumors that are early-responding tissues, while low ratios (1-5 Gray) represent late-responding tissues.
The Alpha/Beta ratio describes how sensitive a specific tissue is to the size of the dose per fraction.
What it means:
- High Alpha/Beta (approximately 10 Gy): Typical for most cancers and “early-responding” tissues (like skin). These are more sensitive to the total dose given over time rather than the size of each individual daily dose.
- Low Alpha/Beta (approximately 1.5 to 3 Gy): Typical for “late-responding” tissues (like the rectum or bladder) and, uniquely, prostate cancer.
Significance in Prostate Cancer: Most cancers have a high alpha / beta ratio, but prostate cancer has an unusually low ratio (estimated between 1.5 Gy and 2 Gy). This means prostate cancer is more sensitive to larger doses per fraction than to many small doses.
The concept of “Therapeutic Gain”: Because the prostate cancer’s ratio (approximately 1.5 Gy) is potentially lower than that of the surrounding healthy rectum (approximately 3 Gy), we can use “hypofractionation” (fewer, larger doses). This theoretically kills more cancer cells while sparing the rectum better than traditional methods.
Abscopal Effects And Synergistic Effects Of Radiotherapy
Radiotherapy (RT) in prostate cancer is increasingly recognised not only for local tumor control but also for its ability to activate the immune system, leading to the abscopal effect (regression of distant, non-irradiated tumors) and synergistic effects when combined with immunotherapy.
While prostate cancer is typically considered an immunologically “cold” tumor—meaning it has low immune cell infiltration—radiation can help convert it into a “hot” tumor, allowing for better systemic response, particularly in metastatic castration-resistant prostate cancer (mCRPC).
The Abscopal Effect in Prostate Cancer
The abscopal effect occurs when localised radiotherapy focused locally to the prostate initiates a systemic anti-tumor immune response, causing untreated remote metastatic lesions to regress.
Radiotherapy (RT) of the primary prostate tumor in oligometastatic prostate cancer can theoretically trigger an abscopal effect, where localized radiation to the prostate induces an immune-mediated shrinkage of distant, untreated metastases. While technically possible, this effect is rarely observed clinically and not the sole mechanism behind treatment success, though it is a key rationale for using Radiotherapy to treat the primary site to improve survival outcomes.
Recent developments focus on combining radiotherapy with immunotherapies to potentially increase the frequency of this response.
Limitations: Prostate tumors often suppress this immune response. Studies show that Radiotherapy alone is usually insufficient, requiring combination with other agents to overcome immunosuppression.
Synergistic Effect of Radiotherapy (RT) and Immunotherapy (iRT)
The combination of RT (Radiotherapy) with immune checkpoint inhibitors (ICIs) like anti-PD-1 (e.g., pembrolizumab) or anti-CTLA-4 (e.g., ipilimumab) is designed to unleash the immune system.
Synergy in mCRPC (metastatic castrate resistant prostate cancer): Combining radiotherapy with ICIs has shown that radiation acts as an “in situ vaccine,” enhancing the visibility of prostate cancer cells to the immune system.
Optimal Timing / Dosing: Preclinical data suggest that fractionated RT (e.g., 8 Gy x 3) is more effective than single high-dose fractions for inducing the abscopal effect when combined with immunotherapy.
Challenges and Future Directions
Despite promise, the “cold” microenvironment of prostate cancer remains a barrier. The future of this approach focuses on:
- Individualized Treatment: Tailoring radiation dose and fractionation to the patient’s immune status.
- Novel Combinations: Utilizing bispecific T-cell engagers or CAR-NK cells alongside RT to convert cold tumors to hot.
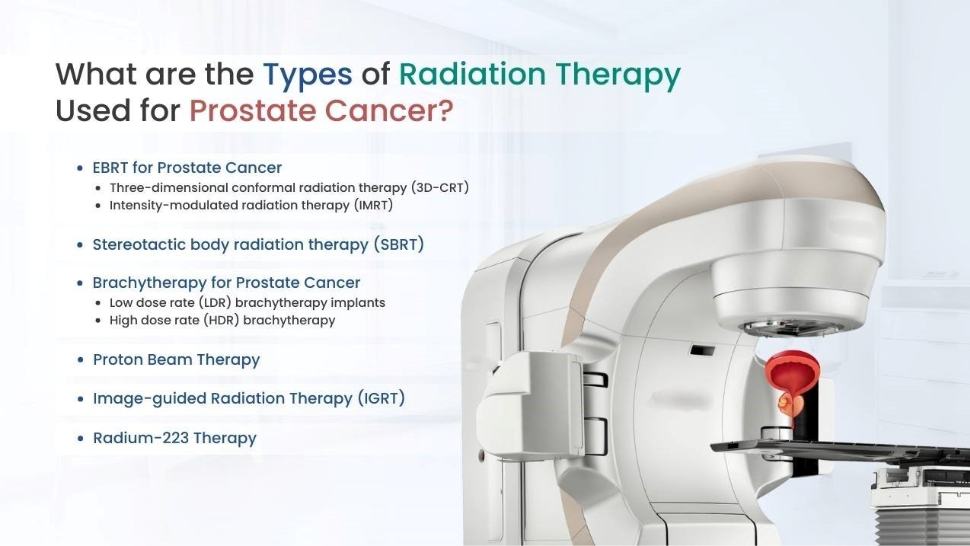
Types Of Radiotherapy Delivery For Prostate Cancer Treatment
adiotherapy can be delivered to the prostate in various approaches:
- EBRT (External Beam Radiotherapy) – radiation beams are directed from outside the body toward the prostate.
- Brachytherapy – Involves placing radioactive materials directly inside the prostate, either by using radioactive seeds (low dose rate / LDR brachytherapy) or by inserting high-activity radioactive source (usually Iridium-192) is inserted into the tumor via catheters or applicators for a few minutes (high dose rate / HDR brachytherapy).
- Combination of EBRT and brachytherapy boost – highly effective treatment for intermediate and high-risk prostate cancer, providing superior biochemical control and survival compared to EBRT alone.
- Systemic Radiotherapy (Radiopharmaceuticals):
- Targeted Radionuclide Radioligand Therapy: A type of systemic radiation, such as 177 Lu-PSMA-617, binds to Prostate-Specific Membrane Antigen (PSMA) on cancer cells, releasing radiation directly to the tumor site. It is a targeted radioligand therapy for PSMA-positive metastatic castration-resistant prostate cancer (mCRPC) that has spread despite prior treatment. It delivers radiation directly to prostate cancer cells, killing them while sparing healthy tissue, significantly extending survival and reducing progression in advanced cases. Since it targets the PSMA protein on the surface of prostate cancer cells, rather than just the bone, it allows to treat both bone and visceral (soft tissue) metastases. Patients must have a positive PSMA Pet / CT scan to confirm PSMA expression.
- Targeted Alpha Therapy: Radionuclide therapies, including Radium-223 (for bone metastases) or Actinium-225, are designed to attack cancer throughout the body. Radium-223 (Xofigo) has largely supplanted strontium and samarium because it is the only bone-targeting agent shown to extend overall survival, in addition to reducing skeletal-related events. It is used for treatment of mCRPC (metastatic castrate resistant prostate cancer) with symptomatic bone metastasis with no visceral organ metastasis. Actinium-225 PSMA-617 has been used for patients with mCRPC who have progressed after Lutetium-177 therapy.
- Bone-seeking beta-emitting radionuclides: Strontium-89 and Samarium-153 are historically significant targeted radionuclide therapies used for the treatment of metastatic castration-resistant prostate cancer (mCRPC) that has spread to the bones. Unlike Radium-223, Strontium-89 and Samarium-153 have no proven survival benefit as single agents in mCRPC patients. Their use is often associated with myelotoxicity (bone marrow suppression), which can limit repeated dosing.
- Rhenium-186 and Rhenium-188 are beta-emitting radioisotopes used primarily for palliative care in advanced prostate cancer, specifically to relieve pain from bone metastases. Research explores their use alongside chemotherapy to enhance tumor destruction
Radiotherapy for prostate cancer in general practice is usually delivered by External Beam Radiotherapy (EBRT), brachytherapy or a combination of both.
EBRT (EXTERNAL BEAM RADIOTHERAPY)
External Beam Radiation Therapy (EBRT) for prostate cancer is a non-invasive, commonly curative treatment that uses a machine (linear accelerator) to direct high-energy X-ray beams (photons) at the prostate from outside the body. It destroys cancer cells or stops them from growing, often used for localised cancer or to reduce symptoms.
EBRT is an effective alternative for patients who cannot undergo surgery, though it may require long-term management of side effects
Common Types of EBRT:
3D Conformal Radiation Therapy (3D-CRT): Shapes radiation beams to match the prostate, though often surpassed by IMRT
IMRT (Intensity-Modulated Radiation Therapy): Highly precise, altering the intensity of beams.
SBRT / SABR (Stereotactic Body Radiation Therapy): Delivers higher doses over fewer sessions (ultrafractionation).
VMAT (Volumetric Modulated Arc Therapy): Advanced, fast-acting form of external-beam radiation therapy (also known as RapidArc) that delivers highly conformal radiation doses to tumors while minimizing exposure to surrounding healthy tissues. It uses a linear accelerator that rotates 360 degrees around the patient, varying the speed, beam shape, and dose rate during treatment.
Image-Guided Radiation Therapy (IGRT): Imaging (like MRI or CT) is used before every session to guide the beam, adjusting for prostate movement.
Proton Beam Therapy: Uses protons instead of X-rays (photons), allowing the radiation to stop at the border of the tumor (the Bragg Peak effect), reducing exposure to healthy tissue.
Carbon ion therapy (CIRT): Carbon ions are even heavier than protons. They have a lower Alpha / Beta ratio sensitivity and a higher Relative Biological Effectiveness (RBE). CIRT is being investigated for “radioresistant” prostate cancers (those that do not respond well to standard radiation) because the carbon ions cause more complex, lethal DNA damage that the cancer cells cannot repair.
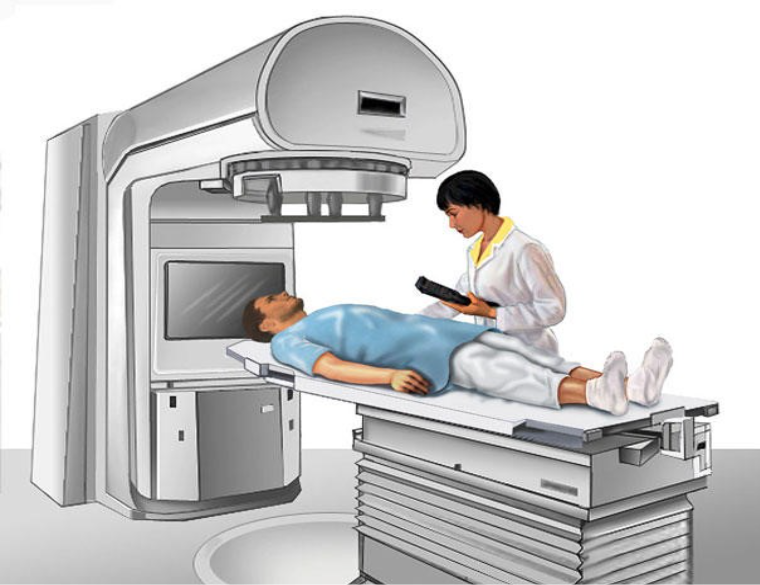
EBRT delivered by a linear accelerator
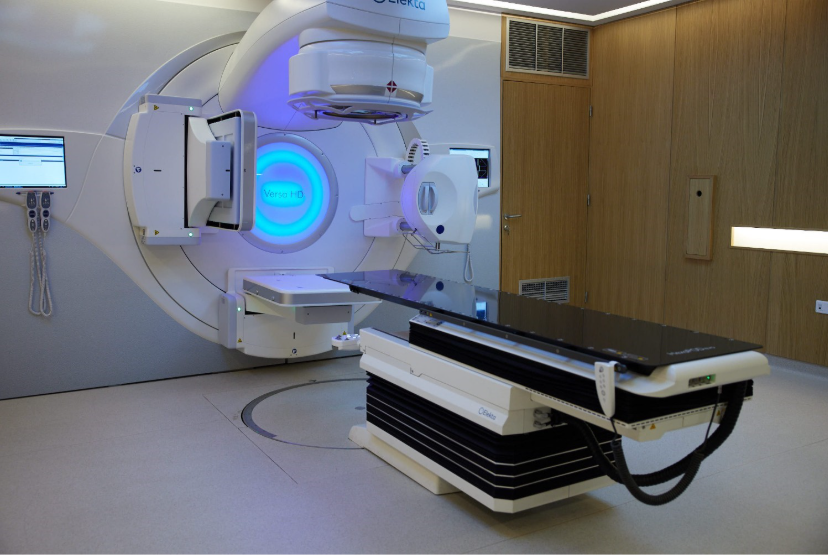
The Elekta Versa HD™ Linear Accelerator for stereotactic radiosurgery
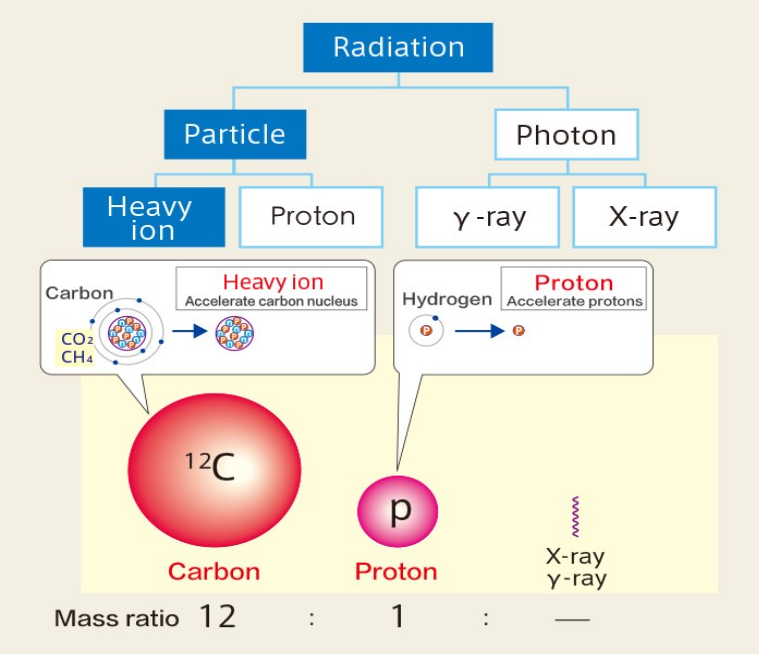
Therapeutic radiation is categorised into either a photon beam, or a particle beam. A photon beam is a kind of electromagnetic wave, and a particle beam is made up of high-energy ions. The former includes X-rays and gamma rays, and the latter includes protons and heavy ions.
Brachytherapy In Prostate Cancer
Brachytherapy plays a crucial role in treating localised prostate cancer, often providing cure rates comparable to radical prostatectomy while offering a better toxicity profile.
The two main types – Low-Dose-Rate (LDR) and High-Dose-Rate (HDR) brachytherapy – differ significantly in dose delivery method and risk group suitability.
It is highly effective for localized cancer, allowing high doses (e.g., 125-145 Gy for LDR; 10.5 Gy X 3 for HDR) to be delivered specifically to the prostate.
Types And Modalities Of Brachytherapy
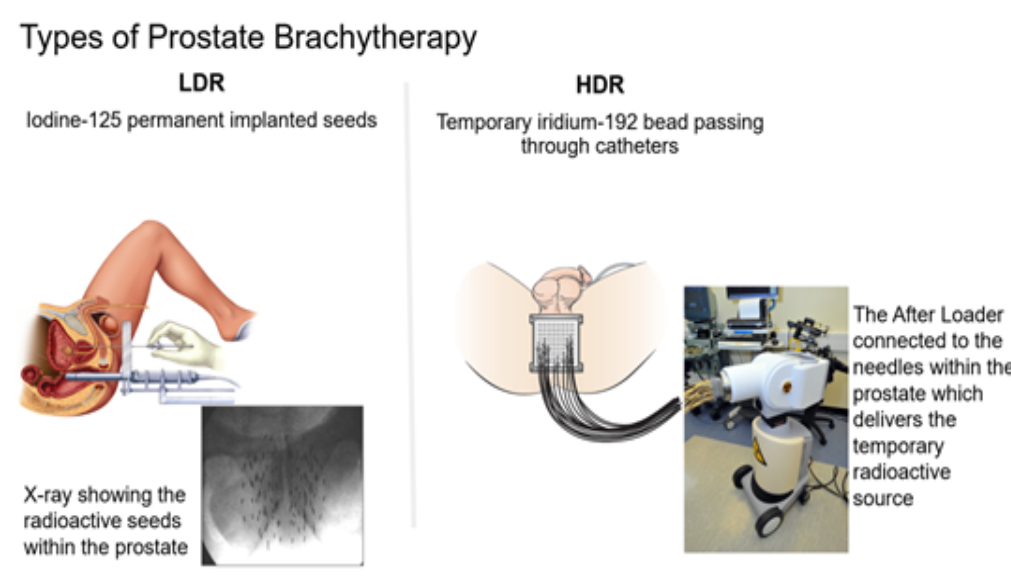
Low-Dose-Rate (LDR) Brachytherapy (Permanent Seed Implants):
Radioactive seeds (e.g., Iodine-125, Palladium-103) are permanently implanted into the prostate. They release radiation over several months, with maximum activity in the first few weeks.
It is usually reserved for low-risk and favorable intermediate-risk cancer.
High-Dose-Rate (HDR) Brachytherapy (Temporary Implants):
Thin, hollow tubes (catheters) are inserted into the prostate under anesthesia, through which a high-activity source (usually Iridium-192) is passed to deliver high-dose radiation in a few minutes, then removed.
This is also often used in combination as a “boost” in combination with External Beam Radiation Therapy (EBRT) for high-risk prostate cancer patients.
Focal Brachytherapy: An emerging modality that targets only the cancer lesion (“index lesion”) within the prostate, aiming to reduce toxicity further.
Side Effects of brachytherapy
Temporary urinary symptoms (frequency, burning, urgency) and occasional erectile dysfunction are the most common, generally appearing a few weeks after treatment.
Role And Risk Group Suitability For Brachytherapy In Prostate Cancer
Low-Risk Prostate Cancer: Primarily treated with LDR monotherapy (sole treatment). HDR monotherapy is also used.
Intermediate-Risk Prostate Cancer: Treated with either LDR or HDR monotherapy in favorable cases, or as a “boost” combined with External Beam Radiation Therapy (EBRT) for unfavorable cases.
High-Risk Prostate Cancer: Usually treated with combined modality therapy (EBRT + HDR or LDR boost) + androgen deprivation therapy (ADT).
Salvage Brachytherapy: Used when the cancer recurs after initial EBRT
Both brachytherapy and External Beam Radiation Therapy (EBRT) are highly effective for treating prostate cancer, with the “better” option depending heavily on risk group and patient factors.
Brachytherapy (internal) is often preferred for low-to-intermediate risk due to higher, more targeted dosing, while EBRT is often better for high-risk, large prostates, or when lymph nodes need treatment.
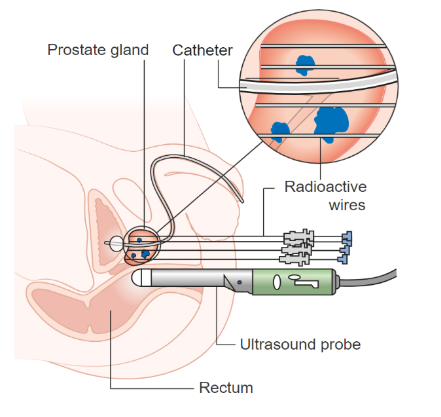
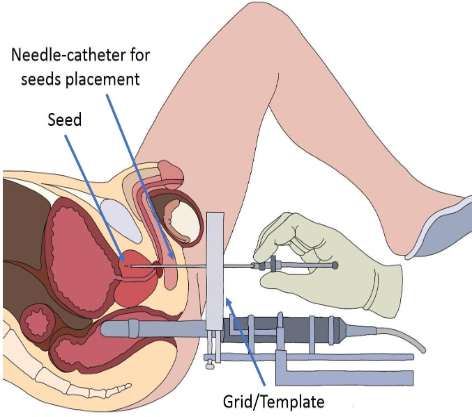
Brachytherapy set-up
Key Considerations
Combination therapy: For high-risk prostate cancer, combining both (EBRT + Brachytherapy boost) is considered superior for survival and controlling disease compared to either alone.
SBRT (Stereotactic Body Radiotherapy): A type of specialized, high-dose EBRT that is gaining favor, with studies showing similar success rates to brachytherapy with fewer urinary issues for some patients.
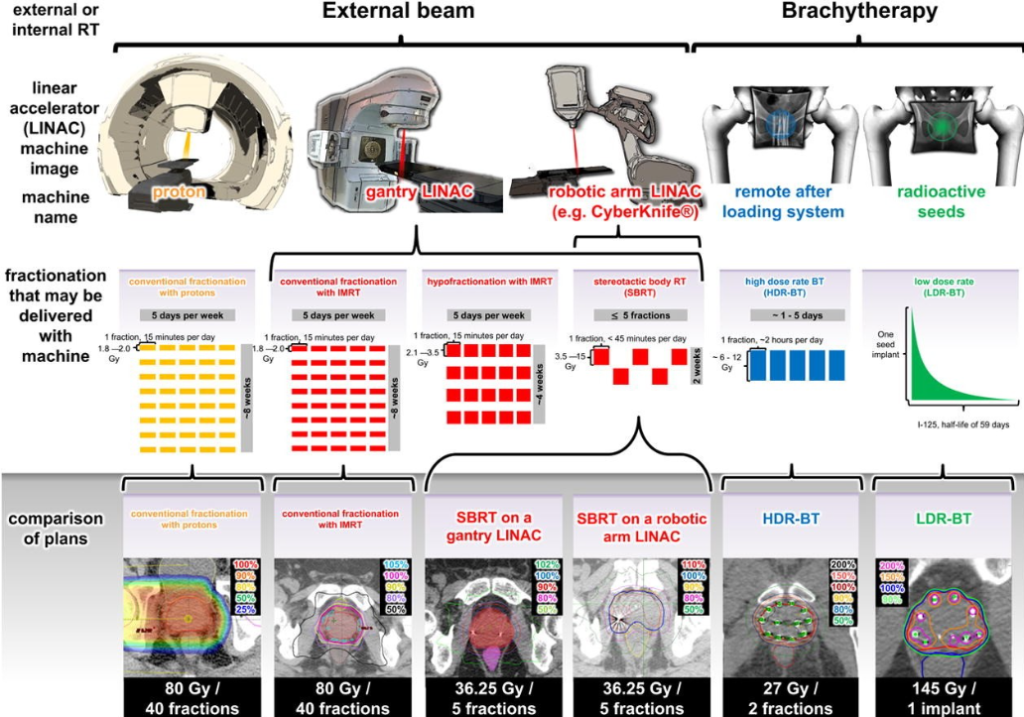
Comparison of EBRT and Brachytherapy plans
Verdict in 2026:
The verdict on the role of radiotherapy in prostate cancer centers on personalised treatment through refined patient selection, the shift toward shorter, high-precision techniques, and its integration with advanced systemic therapies.
The focus in 2026 is on using advanced imaging (like PSMA PET/CT) to personalise radiotherapy, aiming to treat only when necessary and to tailor the intensity of radiation.
While radiotherapy remains a curative cornerstone, its combination with other treatments is becoming more targeted, particularly in reducing unnecessary hormone therapy for certain patients.
Based on findings presented at the 2026 ASCO Genitourinary Cancers Symposium and recent 2026 literature, the consensus on prostate cancer radiotherapy is that while brachytherapy boosts provide superior biochemical control, they do not necessarily prolong overall survival (OS) compared to modern, highly precise external beam radiotherapy (EBRT) techniques, particularly in the long term.
The “best” modality is increasingly personalized based on risk profile and patient preference, with high-intensity EBRT (SBRT) providing strong competition to brachytherapy for intermediate risk, while HDR brachytherapy remains a powerful, albeit more intensive, tool for high-risk disease.
SBRT Emerging as the New Standard:
SBRT is increasingly favored for its convenience (5 treatments over 1 week) with 5-year biochemical progression-free survival rates of 95% for low-risk, 84% for intermediate-risk, and 81% for high-risk localised patients.
The PACE-B trial, a phase 3, international, randomised trial confirms SBRT (fewer, higher-dose sessions) is effective, reducing treatment to only 5 sessions with minimal impact on daily life.
The 10-year update of the HYPO-RT-PC open-label, randomised, phase 3, non-inferiority trial comparing ultra-hypofractionated (SBRT) and conventionally fractionated radiotherapy in intermediate-to-high-risk localised prostate cancer confirms the non-inferiority of the ultra-hypofractionated radiotherapy regimen compared with the conventionally fractionated, with similar toxicity profiles.
With this, as of early 2026, the consensus is that stereotactic body radiotherapy (SBRT) is established as a standard, highly effective, and low-toxicity option for localised prostate cancer, often preferred for reducing hospital visits.
Targeted boosting:
Evidence supports using MRI-visible “boosts” for high-risk patients, while long-term studies affirm ultra-hypofractionation (fewer, higher-dose sessions) maintains comparable survival with less toxicity.
The FLAME prostate trial (Focal Lesion Ablative Microboost in Prostate Cancer) a randomized phase III trial, found that delivering a high-dose focal boost to MRI-visible tumors significantly improves 10-year biochemical disease-free survival (bDFS) compared to standard radiation therapy alone, without increasing toxicities.